How Lab Created Diamonds are Made
How are lab created diamonds made?
Lab-grown diamonds, hailed for their striking similarity to natural diamonds, emerge not from the depths of the Earth, but from the realm of cutting-edge technology within controlled environments. Curious if these diamonds pass muster with diamond testers? The answer is a resounding yes! Lab-grown diamonds indeed yield positive results when subjected to diamond testing methods.
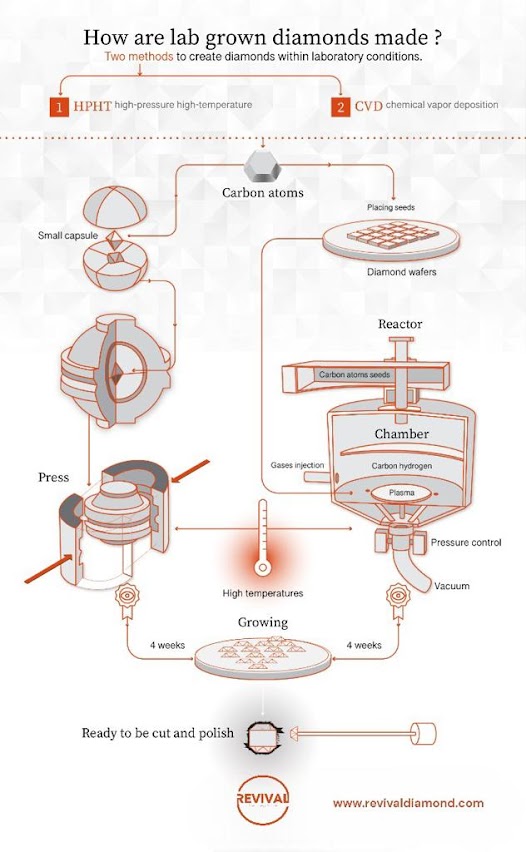
Delving into the production process, two main methodologies stand out: High Pressure High Temperature (HPHT) and Chemical Vapor Deposition (CVD). These techniques meticulously mirror the Earth's natural processes, fashioning exquisite diamonds within controlled lab settings.
High Pressure High Temperature Method (HPHT)
Initially developed in the 1950s for industrial purposes, the HPHT diamond production process has found its stride in crafting lab-grown diamonds. Notably, this method can also enhance the clarity and color of mined diamonds.
So, how does the HPHT process unfold? By replicating the temperature and pressure conditions found deep within the Earth's interior where natural diamonds evolve over billions of years. Unlike the lengthy natural formation process spanning 1-3 billion years, the HPHT technique ingeniously accelerates diamond creation to mere weeks!
The HPHT process employs three primary manufacturing techniques: the split sphere (BARS) press, the cubic press, and the belt press. Each method envelops a tiny diamond seed within carbon and subjects it to high pressure and temperature for several days. As the carbon liquefies, it fuses with the seed, gradually giving rise to a full-fledged diamond.
Chemical Vapor Deposition (CVD)
Enter the world of CVD, a process reminiscent of how diamonds arise in interstellar gas clouds. This recently developed technology, a hallmark of the 1980s, employs hydrocarbon gas mixtures to synthesize diamonds. Notably, the CVD process boasts cost-efficiency, harnessing less energy than HPHT.
How does CVD work its magic? A sliver of diamond seed is carefully heated to approximately 800°C within a sealed container, subsequently infused with carbon-rich gases, including methane. Microwaves, lasers, and other tools step in to ionize the gases and generate plasma. This ionization-induced gas breakdown facilitates the merging of carbon with the diamond seed, layer by layer, ultimately culminating in the formation of a diamond.
Intriguingly, lab-created diamonds, while birthed through technological prowess, bear the same chemical composition, physical traits, and visual allure as their naturally occurring counterparts. As you marvel at their brilliance, remember that these diamonds are a testament to human ingenuity and a sustainable path forward.
How is the production of CVD diamonds accomplished?
Initially, a tiny piece of diamond seed is subjected to temperatures of approximately 800 degrees Celsius within a sealed chamber. Subsequently, the chamber is infused with carbon-rich gases like methane.
Following this, microwaves, lasers, and other tools are employed to ionize these gases, resulting in the formation of plasma. The ionization process facilitates the decomposition of the gases, allowing the carbon to combine with the diamond seed. Consequently, this leads to the gradual layer-by-layer growth of the diamond.